Knee Osteoarthritis
Knee osteoarthritis is a degenerative joint condition characterized by cartilage wear, inflammation, and reduced mobility. Regenerative medicine approaches such as umbilical cord–derived mesenchymal stem cells (MSCs) are explored to support cartilage health, reduce inflammatory signaling, and promote joint function under medical supervision.
Introduction to Knee Osteoarthritis
Knee osteoarthritis is a common degenerative joint condition that affects cartilage structure, joint flexibility, and overall mobility. It is often associated with chronic inflammation, stiffness, and discomfort during daily movement. In regenerative medicine, umbilical cord–derived mesenchymal stem cells (MSCs) are increasingly explored as a supportive therapy aimed at promoting cartilage environment health, regulating inflammatory responses, and assisting natural joint recovery pathways under medical supervision.
In Turkey, stem cell therapy for knee osteoarthritis is performed using GMP-certified laboratory-processed cells combined with individualized treatment protocols. Because MSC therapy focuses on biological signaling and tissue support rather than surgical replacement, it is considered by many patients seeking non-invasive regenerative options. Treatment suitability is determined through medical consultation and clinical evaluation.
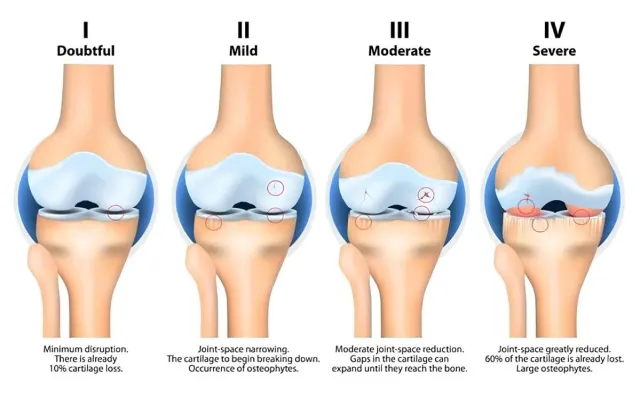
What Is Knee Osteoarthritis
Knee osteoarthritis is a progressive joint disorder characterized by cartilage wear, joint space reduction, inflammation, and decreased range of motion. Factors such as aging, previous injuries, excess weight, and repetitive stress can contribute to its development. Symptoms may include stiffness, swelling, and discomfort during walking or physical activity. Early assessment and supportive care play an important role in maintaining joint function and mobility.
How Stem Cells May Support Cartilage
Mesenchymal stem cells function through paracrine signaling, meaning they release bioactive molecules that influence the surrounding cellular environment. Instead of replacing cartilage directly, MSCs are studied for their ability to regulate inflammation, support tissue repair pathways, and encourage natural cellular communication involved in joint maintenance. This biological mechanism is why stem cell therapy is described as regenerative support rather than structural replacement.
Why Umbilical Cord MSCs
Umbilical cord–derived MSCs are considered a preferred source in regenerative medicine due to their youthful cellular profile, higher proliferation potential, and lower immunogenic response compared to adult tissue sources. These cells are ethically obtained from voluntary donations after childbirth and processed in GMP-certified laboratories that follow international sterility and quality standards. Many international patients search for umbilical cord stem cell therapy in Turkey because of these laboratory and regulatory advantages.
Combination With PRP and Exosomes
In some treatment protocols, stem cell therapy may be combined with Platelet-Rich Plasma (PRP) or exosomes therapy to enhance biological signaling pathways. PRP uses the patient’s own growth factors to support tissue recovery, while exosomes act as cellular messengers that influence inflammation control and repair communication. Combination approaches are determined by physicians based on medical suitability and individual treatment goals.
Treatment Process
A typical knee stem cell therapy journey includes:
1
2
3
4
5
6
Treatment duration and protocols vary depending on individual clinical evaluation.
Safety & Eligibility
Eligibility for knee stem cell therapy depends on medical history, diagnostic reports, and physician assessment. Treatments are performed under clinical supervision using laboratory-tested biologic materials and sterile medical protocols. While regenerative medicine continues to evolve, transparency, realistic expectations, and professional evaluation remain essential components of patient safety.
FAQs
Free Online Medical Consultation
Speak with a medical advisor and receive a personalized evaluation based on your medical reports. No obligation — professional guidance to help you understand available regenerative treatment options.
Book Free Consultation